The global demand for South Australian mineral resources remains high and major international companies, mid-caps and juniors continue to operate and invest in the South Australian resources industry.
Investors and other stakeholders are presented with significant opportunities and benefits across the industry value chain.
Strategic Hydrogeological Framework
The strategic hydrogeological framework is a joint collaboration between the Department for Energy and Mining (DEM) and the Department for Environment and Water (DEW).
It forms part of the Water and Infrastructure Corridors Initiative (WICI), aimed to address water access and productivity constraints in the state.
DEM has led the development of the strategic hydrogeological frameworks for the Braemer province, the Stuart Shelf region, and for the Northern Eyre Peninsula.
The frameworks examine the potential for groundwater resources to mitigate water constraints to mining development. They comprise the most contemporary and comprehensive data and information available on groundwater in each region, and include analyses of projected water demand and the capacity of the groundwater resources to support this demand.
By improving our knowledge of groundwater resources, we aim to minimise risks and create greater certainty to investors seeking to progress resources projects within the arid regions of South Australia.
Northern Eyre Peninsula strategic hydrogeological framework
The goal of this information is to ultimately unlock the development opportunities of the Eyre Peninsula and guide regional development of water infrastructure to underpin jobs, business activity and royalty revenue to support the delivery of government services for all South Australians.
Northern Eyre Peninsula mineral deposits include:
- Magnetite - Middleback Ranges (also hematite resources); Central Eyre Iron Project; additional iron ore resources identified
- Graphite - Uley project; Siviour; Campoona Shaft; Kookaburra Gully; additional graphite resources identified
- Potential for uranium, rare earth elements and kaolin, supported by renewable energy generation and hydrogen production
South Australia Strategic Hydrogeological Framework - Northern Eyre Peninsula (PDF 8.1 MB)
Stuart Shelf Region strategic hydrogeological framework
This framework covers the Stuart Shelf, part of the larger Gawler Graton and a world-renowned copper province.
Stuart Shelf Region mineral deposits include:
- Copper-gold deposits (operating) - Olympic Dam; Carrapateena; Prominent Hill
- Several other copper deposit discoveries, including Oak Dam
South Australia Strategic Hydrogeological Framework - Stuart Shelf Region (PDF 9.6 MB)
Braemar Province strategic hydrogeological framework
This framework covers the Braemar Province, a remote part of the state that straddles the Barrier Highway with limited social infrastructure but an identified potential for resource development.
Braemar Province mineral deposits include:
- Gold - White Dam gold mine (operating)
- Uranium - Honeymoon uranium mine (operating)
- Magnetite - several discoveries
- Copper - several discoveries
South Australia Strategic Hydrogeological Framework - Braemar Province (PDF 5.7 MB)
Fraser Institute Annual Survey of Mining Companies 2022
South Australia has again ranked top ten globally in the 2022 Fraser Institute’s annual survey of mining companies.
Download the report: Fraser Institute Annual Survey of Mining Companies 2022 (PDF 1.1 MB)
Key indices (world rankings)
- South Australia 9th in overall ranking
This takes into consideration both mineral and policy perceptions, and rates regions based on their geological attractiveness, as well as effects of government policy and attitude toward exploration investment. - South Australia ranked 3rd in policy perception index
This measures a jurisdiction's policy practices, including regulatory environment, taxation and political risk. South Australia increased its score by 12.9 points and significantly improved its ranking from 2021, when it came in 16th. - South Australia ranked 1st for the quality of our geological databases
Our databases provide world class data and services, and open up opportunities for exploration and mining. Our demonstrated prospectivity for in-demand and critical commodities such as copper, graphite, magnetite, heavy mineral sands and Rare Earth Elements makes South Australia an attractive destination for explorers.
Our demonstrated prospectivity for in-demand and critical commodities such as copper, graphite, magnetite, heavy mineral sands and Rare Earth Elements makes South Australia an attractive destination for explorers.
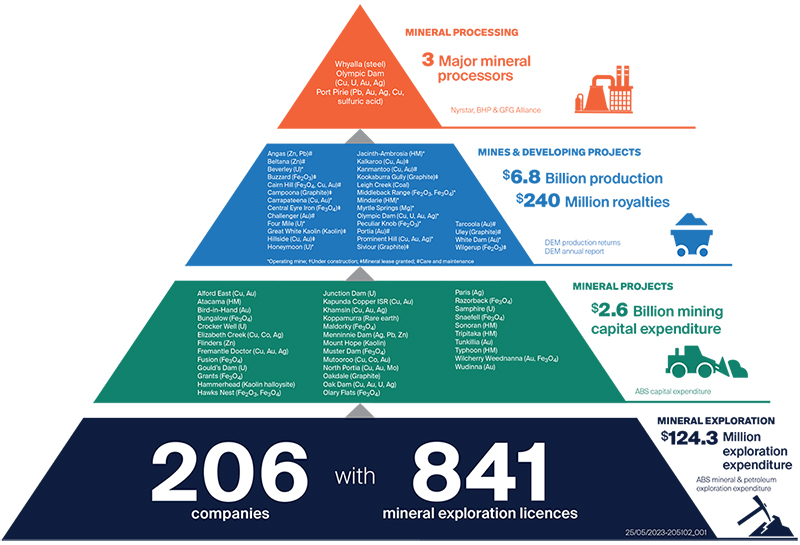
Mineral industry project pipeline
South Australia’s Minerals industry project pipeline shows the breadth of projects progressing through the Minerals Industry Value Chain with benefits for our State.
Further information and spatial location of these projects can be found on South Australia’s Resources Information Gateway (SARIG)
Download South Australia Mineral Industry Project Pipeline (PDF 77 KB)
More about South Australia's minerals industry value chain
Related information
Mineral Exploration in South Australia 2021: Commodity report (PDF 67.4 MB)
South Australia developing projects (PDF)
South Australia mines and advanced projects (PDF)
South Australia major exploration models (PDF)
Current mineral exploration licences and applications in SARIG
More information
Email: DEM.customerservices@sa.gov.au
Phone: +61 8 8463 3000